The ForeseeHome AMD Monitor
Zum Amsler-Test
Source: www.notalvision.com
The ForeseeHome™ AMD Monitor
The ForeseeHome™ AMD Monitor is a breakthrough in the monitoring of age-related macular degeneration (AMD). It is the first FDA cleared ophthalmic device linking patients and doctors, giving doctors access to valuable patient data through a secure online connection. Designed for frequent monitoring between eye exams in the physician's office, the ForeseeHome™ AMD Monitor provides personalized monitoring for patients at risk of vision loss from the devastating effects of wet AMD.
ForeseeHome™
The ForeseeHome™ AMD Monitor is the first ophthalmic device linking patients and doctors between eye exams for ongoing monitoring of age-related macular degeneration (AMD). This telemedicine based system gives doctors access to valuable patient data through a secure online connection, allowing frequent and personalized monitoring of patients at risk of vision loss from the devastating effects of wet AMD.
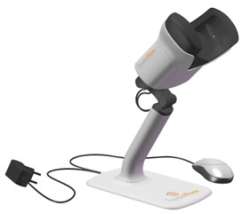
The ForeseeHome™
Photo: Notal Vision
Foresee Home Indications for Use
The ForeseeHome™ AMD Monitor is indicated for use as follows:
The Foresee Home is intended for use in the detection and characterization of central and paracentral metamorphopsia (visual distortion) in patients with age-related macular degeneration, as an aid in monitoring progression of disease factors causing metamorphopsia including but not limited to choroidal neovascularization (CNV). It is intended to be used at home for patients with stable fixation.
The Foresee Home™ is not intended to diagnose; diagnosis is the responsibility of the prescribing eye-care professional.
The appropriate type of patients are intermediate AMD as defined by the Age related Eye Disease Study (AREDS), National Eye Institute.1
- The Foresee Home device will not be useful in patients who have unstable fixation.
- The effectiveness of this device has not been evaluated in AMD patients:
- Who have any co-existing retinal or choroidal disease
- Who have significant media opacity precluding a clear view of the macula
- Who have previous surgical or laser treatment within the macular area, or
- Who have a recent non-macular ocular surgery.
1 Age-Related Eye Disease Study Research Group. A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and E, beta caroteen, and zinc for age-related macular degeneration and vision loss: AREDS report no. 8. Arch Ophthalmol 2001; 119: 1417-1436.
Product Features
ForeseeHome™ Device
- Field tested ergonomics and human factors driven design
- Simple plug and play operation
- Automatic eye detection
- Closed hood and head sensors to control for ambient light and distance from display
- Mouse operated
ForeseeHome™ Test
- Built-in interactive self tutorial designed for elderly patients
- Short (~ 3 minutes per eye)
- Sensitive and specific test based on PHP technology
- Test reliability module
ForeseeHome™ Analysis
- State of the art machine learning classification techniques
- Establishment of a baseline pattern of tests
- Exclusion of patients unable to establish a baseline
- Spatiotemporal abnormality maps
ForeseeHome™ Telemedicine
Call center assistance with initial installation
Call center intervention when compliance falls below requested minimum
Privileged on-line access to patient's information according to role (eye care professional, data monitoring center, administrator, patient).
Accessible web report that includes daily test results, trend analysis, spatial maps of interest, frequency of usage
Read more: ForeseeHome (notalvison.com)